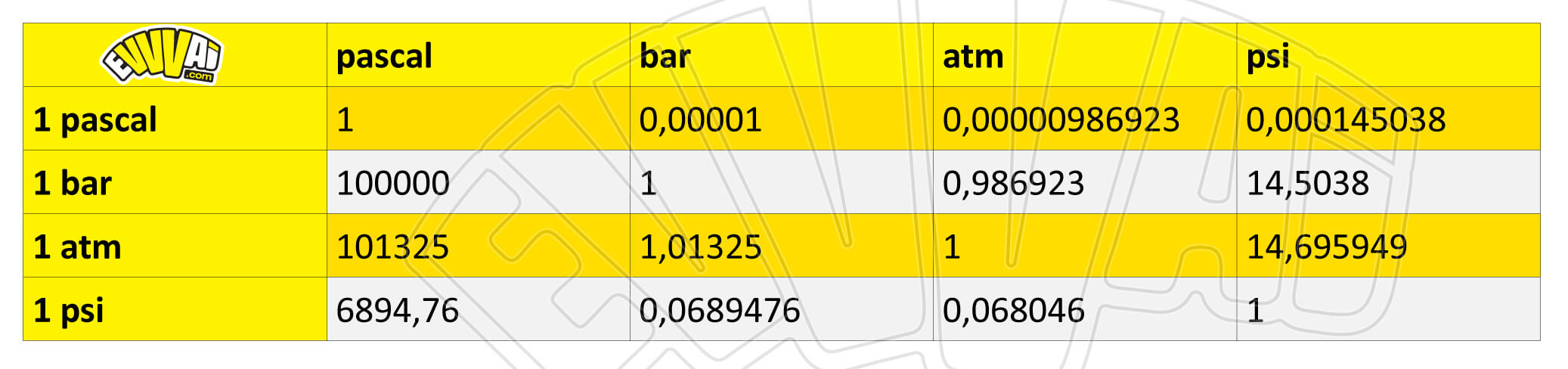
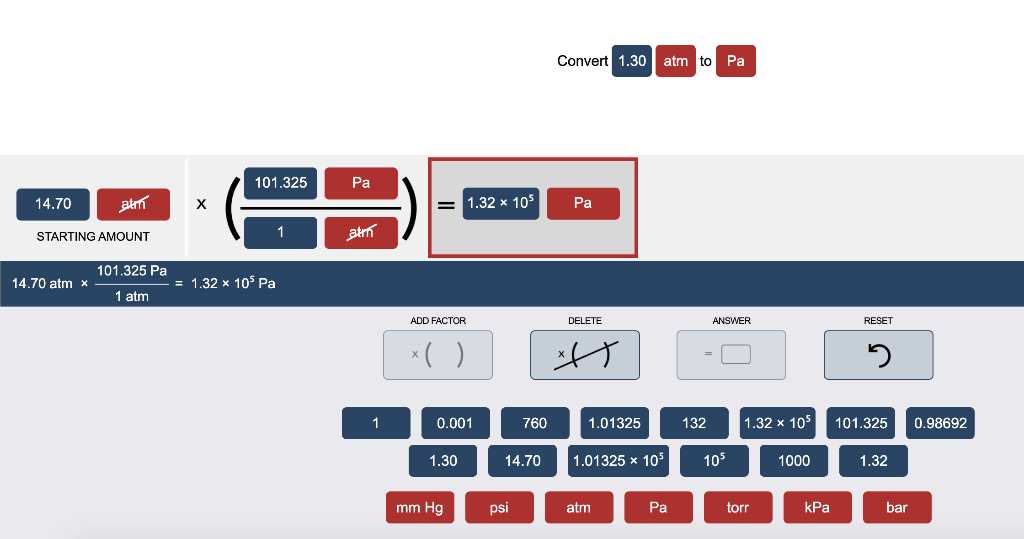
SOLVED: Equalities 1 atm 760 torr (exact) 1 atm 760 mmHg 1 atm 29.92 inHg 1 atm 101325 Pa 1 atm 14.7 psi Equations 9PF TF = Tc + 328F 5PC 5*C

How to convert Atmospheric pressure to Pascal (atm-Pa) and Pascal to Atmospheric pressure (Pa-atm). - YouTube
The guage pressure exerted below a column of water, open to the earth's atmosphere at depth of 10 m is (density of water = 1000 kg/m3, g = 10 m/s2 and 1 atm pressure = 105 Pa)

pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

the SI unit of pressure is the pascal, pa, but in the V-P relationship of gases, it is necessary to be - Brainly.ph



:max_bytes(150000):strip_icc()/GettyImages-124564231-58b5c7d75f9b586046caddb9.jpg)